As discussed in the previous section about the types of cleansers and how they are characterized, true soaps exclusively use one family of surfactants: alkyl carboxylates, which are basically (alkali) salts of fatty acids. While the most common one is sodium stearate, there are many out there. In order to know which ingredient is a soap, you need to be able to recognize what an alkyl carboxylate looks like on an ingredients list.
Keep these points in mind:
- They are almost always two-worded.
- The first word is typically either sodium or potassium. (Notice that these elements are part of the alkaline group of metals on the periodic table; hence the name alkali salts).
- The second word is usually a fatty acid that ends in “-ate.” Commonly used fatty acids are stearic acid (stearate), palmitic acid (palmitate), and myristic acid (myristate). If you aren’t sure, just Google the second word to find out if it’s a fatty acid.
- While this has nothing to do syntax, you should only be concerned if a soap appears in the upper half of the ingredients list, assuming that it’s not too short.
With that out of the way, we can now explore the plethora of surfactants that are commonly used in liquid cleansers, some of which are also used in syndet cleansers.
Surfactants Used in Liquid Cleansers
There are four types of surfactants, which are grouped by the charge of their head groups–something that determines a compound’s interactivity. The carbon chains or backbones are almost always non-reactive. They four types are: anionic, cationic, amphoteric, and nonionic.
The surfactant types in decreasing order of potency are as follows: anionic = cationic > amphoteric > nonionic. Note that this is only a trend, rather than an absolute rule.
Anionic Surfactants
Anionic surfactants have an overall negative charge and lather well. Examples of anionic surfactants include the alkyl carboxylates (yes, soaps are anionic surfactants), ammonium lauryl sulfate, and sodium laureth sulfate. Note that in this case, the identity of the first word typically suggests form, while the second and third words (if there are any) infer function. For example, sodium lauryl sulfate and ammonium lauryl sulfate are equally stripping (function), while the former is less soluble in water than the latter (form)–something that the formulators can compensate by for example, altering the amounts/types of solvents and colorants used. But that difference doesn’t meaningfully affect how well the surfactants remove oil, makeup, and debris from the skin.
Isethionates, phosphates, sulfosuccinates, and sulfonates are also anionic surfactants. Obviously, other sulfates are also anionic. Keep in mind that anionic surfactants represent the most populated and varied group of surfactants. They are varied in the sense that they exhibit widely different degrees of potency. For example, sulfates and sulfonates are significantly more potent than isethionates. Furthermore, newfangled amino-acid based surfactants such as the gluatames, sarcosinates, and glycinates are even more gentle.
Note that there is no research demonstrating that sulfates are carcinogenic. So, rest easily. If anything, I’d venture to guess that a more likely source of sulfate-related carcinogens would come from the byproducts of the production process, rather than from the actual sulfate compounds themselves. But even if that were to be true, the amounts produced would be miniscule or even infinitesimal. Remember, poisons/toxins/carcinogens are dose-dependent (like most things). And really, if you need to worry about a source of carcinogens, worry about the sun. Or overcooked meat and complex carbohydrates. Or smoking…
Cationic Surfactants
Cationic surfactants have an overall positive charge and lather well. Examples of cationic surfactants include benzalkonium chloride and cetrimonium bromide.
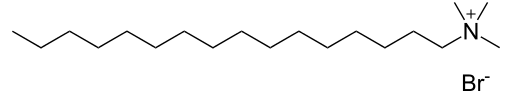
Cetrimonium bromide (a cationic sufactant) is used more for its antibacterial rather than cleansing characteristics.
Cationic surfactants are used less often than anionic ones because:
- They tend to be more expensive;
- They tend to confer stronger antibacterial properties, which can decrease tolerability, thereby widespread use;
- And finally, they tend to be poorly tolerated due more so to their allergic potential rather than their cleaning or “stripping” potency.
Amphoteric Surfactants
Amphoteric surfactants have an overall neutral charge and lather well. Examples of amphoteric surfactants include cocamidopropyl betaine and sodium lauroamphoacetate. It has been shown that combining milder amphoteric surfactants with the traditionally more potent anionic surfactants, can mitigate the overall level of potency. Note that, you’ll occasionally see amphoteric surfactants described as being zwitterionic. While similar and overlapping in characteristics, these two terms do not describe identical concepts. Amphoteric means that a compound can either act as an acid or a base, while zwitterionic means that a compound has an overall neutral (electrical) charge, with a positive and negative charge within the molecule. Since most amphoteric surfactancts are zwitterionic and vice versa, the terms can be used interchangeably when speaking in the vernacular. But it’s technically incorrect to do so. For example, water is amphoteric, since it can act as a base (hydroxide ion) or an acid (hydrogen or hydronium ion). However, water is not zwitterionic, since there are no electrical charges. While it is a neutral compound, because the neutrality is achieved with partial charges (dipoles), it does not qualify as a zwitterion.
Okay, that was a pretty long tangent. Let’s take it a step further? Guess what rhymes with zwitter? Twitter! Lame, I know.
Nonionic Surfactants

Notice how decyl glucoside has no electrical charge in its head group? That makes it a nonionic surfactant.
Nonionic surfactants have no charge and tend to not lather well. Examples of nonionic surfactants include decyl glucoside, cocamide MEA, and polysorbate-20. Interestingly enough, certain nonionic surfactants such as cocamide DEA, were shown to be more irritating than some of the commonly crucified anionic surfactants such as sodium laureth sulfate. This further demonstrates that evaluating cleansers based on a set of rules, is hardly reliable. But it’s better than nothing, right?
Takeaway Message for Surfactants
When I originally brainstormed how to structure this post, I planned on giving a brief overview of each family of surfactants (i.e. sulfates, sarcosinates, etc.). However, I ultimately decided against that because it’s far too provincial to only look at the surfactants in a cleanser when evaluating if one is appropriate for your skin type. Furthermore, it over-complicates things; there are just too many exceptions to any rule or methodology that can be conceived. For example if I were to say that sulfates were the most potent, I would have to make a distinction between sodium lauryl sulfate and sodium laureth sulfate. And while sulfonates tend to be less potent than sulfates, sodium C14-16 olefin sulfonate is more potent than sodium laureth sulfate. Therefore, I’d have to write an asterisk there or something. If done this way, I’d basically have asterisks everywhere, which pretty much defeats the purpose of having a ranking system.
The only ranking system that would be valid is if I were to rank every individual surfactant in order. I would have to find out each surfactant’s CMC. To the best of my knowledge, there is no publicly-available all-inclusive database with this information. Furthermore, a surfactant’s CMC may not even be entirely responsible for a surfactant’s potency. Recent studies suggest that micelle size, surfactant length, and several other aspects can also affect potency. Not to mention there are different ways to measure CMC; each method has its pros and cons. However, newer methods of determination may simplify and provide more consistent answers in the future.
Still, even if such a database were to exist, it would be an incredible waste of my time and yours to put much faith in it, since so many other aspects including the concentrations of the surfactants, emollients and humectants used; as well as an individual’s skin type and skin care routine, etc… will affect the final outcome. Nit-picking and meticulousness are not virtues in this situation.
Therefore, the purpose of this post is to act as a reference of sorts, and to help you understand some of the terms and misconceptions that are thrown around in the dermatological blogosphere. You shouldn’t really concern yourself with what surfactants are present in a cleanser, except when it is close to the front of the ingredients list or if you are sensitive to one or more of them. I admit that on a theoretical level, cleanser evaluations are the least reliable. It’s why I don’t like reviewing cleansers. Consequently, I will NOT be recommending a list of cleansers at the end of this series. Fear not however, there will be a general article that discusses how to choose an appropriate cleanser (LINK) at the end of this section/series.







Hm. I am really curious about sodium olefin sulphonate. So, stronger than sodium laureth sulfate, but … less strong than… ? I recently bought 1l and I am planning to use it in my shampoo. I was also under the impression that this is a milder surfactant, and then I read Paula’s Choice’s comments on it (“beware, that is an irritant!!!”) and haven’t found much other evidence . So, if you would have any comments about that it would be awesome!
Posted by Simona | May 25, 2014, 11:04 pmIt’s a combination of of sodium olefin sulfonate’s higher CMC, sulfonate head group, and carbon chain length, that makes it more irritating that SLES, but less so than SLS, though not by much.
Posted by John | May 29, 2014, 10:01 am